The Infection Research Initiative is a robust, comprehensive program to improve our knowledge of cystic fibrosis-related infections, develop safe and effective treatments, and improve current treatments for people with CF. The initiative has already significantly expanded the Cystic Fibrosis Foundation’s infection research portfolio.
Areas of Research Focus
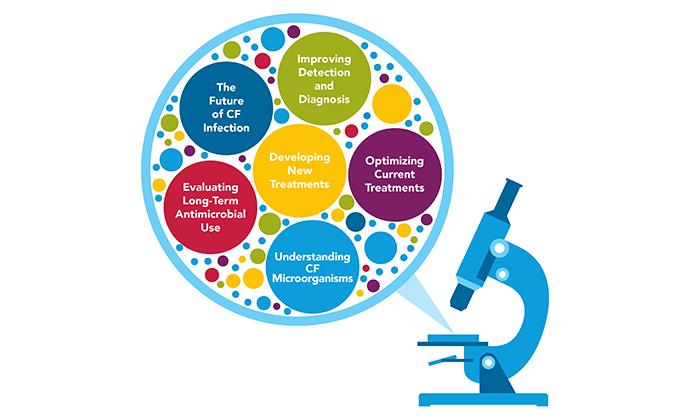
The Infection Research Steering Committee -- made up of CF community members and experts in CF clinical care, research, and drug development -- created a comprehensive research program focused around six key priorities:
1. Improving Detection and Diagnosis
Scientists are focusing on developing new technology to detect microorganisms (bacteria, viruses, fungi, etc.) from cultures that are quicker, more accurate, and less invasive than sputum samples and mouth swabs. Non-culture-based methods are also being explored.
To help patients and clinicians make informed decisions about the best course of treatment, researchers are conducting studies to determine whether to monitor or treat a detected microorganism in the lungs or sinuses. For example, in some cases, early treatment for colonization may be unnecessary, while in others, it may be best to take preemptive action.
2. Understanding CF Microorganisms
To improve ways to prevent and treat infections, researchers are trying to better understand CF microorganisms -- how they get into someone's lungs and how the body's immune system responds, New technologies, such as whole genome sequencing and single-cell sequencing, can be used to map out the entire genetic structure of a bacterium. This mapping can help researchers understand how certain bacteria cause disease and become drug-resistant and help them uncover ways to interrupt these disease-causing processes either with novel antibiotics or non-traditional treatments.
3. Developing New Treatments
The CF Foundation meets with numerous companies every year to identify and accelerate potential therapies, including antibiotics, antivirals, and antifungals. In addition, we are supporting research into new types of antimicrobials, such as bacteriophage or phage therapy, which uses viruses that attack bacteria to treat bacterial infections.
To help move potential treatments quickly through clinical trials in humans, we need to develop models of the CF lungs to use in preclinical studies to determine if a new drug will work in people with CF. However, the lungs of someone infected by cystic fibrosis are unique and difficult to replicate. To overcome that problem, the Foundation supports research into new systems that can replicate the environment of the CF lung. For example, the “lung on a chip” is a microchip that can mimic how the lung responds to different stimuli.
4. Optimizing Current Treatments
To make the most effective use of current therapies, research is being conducted to understand how therapies may interact with other medications and whether people with CF need to adjust their doses or courses of treatment accordingly. We are also supporting research to look at combinations of therapies for infection to determine which combinations are most effective against nontuberculous mycobacteria (NTM), methicillin-resistant Staphylococcus aureus (MRSA), and Pseudomonas.
5. Evaluating Long-Term Antimicrobial Use
People with CF are prescribed antibiotics frequently or may take antibiotics long-term as part of their standard daily care. The CF Foundation is interested in research projects to understand the risks and benefits of antimicrobial use and their related complications and is funding research to mitigate or treat known risks, such as hearing loss.
6. The Future of CF Infections
As we make tremendous strides in developing CFTR modulators and other drugs, it is vital that we begin to understand how these drugs will affect CF infections. Research in this area of focus will help shape clinical care as we prepare for further breakthroughs on our path to a cure. The CF Foundation is supporting the development of two large research studies to understand how infections will evolve as people with CF start on highly effective modulator therapy -- treatment that provides more than a 10 percent increase in FEV1 and at least a 50 percent reduction in pulmonary exacerbations.1
Current Portfolio and Progress Since Infection Research Initiative Launch
Since the Infection Research Initiative launched in 2018, progress is being made to explore new areas of infection research and advance priorities identified by the steering committee and the CF community. Scientists are studying key CF microorganisms across all six areas of research focus.
The current infection research portfolio serves as a snapshot of where we are now and provides a benchmark to demonstrate how far we have come, especially in areas that have been identified by the community as priorities. We are pleased to note progress in areas of priority to the CF community, including research to understand the impact of CFTR modulators on CF infections.
To understand the impact of the modulator Trikafta® on CF infections, the Foundation dedicated a portion of the PROMISE study to increase our understanding of the changes in the microbiome and infection status of people with CF who start on the triple-combination therapy. The PROMISE study will assess the overall impact of Trikafta on people with CF.
As researchers start to focus more on microbial communities and how microbes interact with each other instead of studying them individually, projects that look at multiple organisms have increased. Because microbes are rarely found alone, these studies can help us better understand microbes in a more realistic environment.
Consistent with our goal to fund areas that will directly impact people with CF, there were substantial increases in funding programs developing new treatments, looking at the future of CF infections, and optimizing current treatments. To learn more about this funding, visit the infections section of Research We Fund.
The microorganisms listed below are being studied in the current infection research portfolio. Microorganisms not listed below (e.g., Achromobacter xylosoxidans) have been identified as research gaps and will be addressed by the Initiative. Read more about what we are doing for each microorganism, and visit the Drug Development Pipeline to learn more about new therapies in development or already in use by patients.
- Pseudomonas aeruginosa
- NTM
- Multiple Organisms (more than one microorganism)
- Staphylococcus aureus/MRSA
- Burkholderia cepacia
- Bacteriophage (Phage) Therapy
- Viral
- Fungal
- Stenotrophomonas maltophilia and other gram-negative bacteria
Pseudomonas aeruginosa
Pseudomonas research projects compose almost 40% of the infection research portfolio with a third of that funding going toward developing new treatments, including phage therapy. Since the launch of the initiative, eight new drug development programs targeting Pseudomonas infection were funded, three of which use phage therapy to target Pseudomonas infections.
NTM
Nontuberculous mycobacterium is an emerging pathogen that can be difficult to treat and has become more common recently in people with CF, infecting nearly 10% in 2020. The CF Foundation is supporting two large clinical studies to better understand when to treat someone who cultures NTM and to standardize antibiotic treatment for people with NTM infections. In 2019, two new drug development programs, focused on identifying novel treatments for NTM, were funded. In 2021, another program was funded to develop a portable nitric oxide treatment.
Multiple Organisms
Multiple Organisms encompasses research that includes developing treatments that have the capacity to treat Pseudomonas and NTM (or others), and research into finding ways to treat pulmonary exacerbations that may not be linked to a specific microorganism. The Foundation is expanding support for research projects specifically looking at the microbial mechanisms, including viruses, that drive exacerbations.
Staphylococcus aureus/MRSA
The majority of the portfolio dedicated to Staphylococcus aureus (S. aureus) and MRSA focuses on improving our understanding of how the body may respond to infections from these bacteria. The portfolio also includes research to optimize current treatments and develop new treatments.
Burkholderia cepacia
Infections caused by the 22 bacterial species known as Burkholderia cepacia complex are rare among people with CF, but severe, and there are few treatments available. Since the launch of the initiative, the Foundation has funded a drug discovery program to screen for a compound that could be used to treat these infections.
Bacteriophage (Phage) Therapy
Although it is legal in some parts of Europe, phage therapy, which uses viruses to target difficult-to-treat bacteria, has not been approved by the U.S. Food and Drug Administration for use in the U.S. To date, phage therapy has been primarily used to treat acute infections. Because CF infections are often chronic and microorganisms tend to colonize the CF lung, there are many unique challenges that must be overcome before phage therapy can be considered safe and effective for people with CF. Since the launch of the initiative, the Foundation has funded four studies in phage, including the first ever controlled clinical study of phage in people with CF that can start to help us answer the question of whether phage therapy is a safe and effective way to treat CF infections.
Viral
Research centers on understanding how viral infections affect people with CF, including the role of viruses in CF exacerbations. Scientists are also investigating how viruses may interact with other microorganisms in the CF lung. Because we recognized the need to address the unknowns of the global pandemic for the CF community, the Foundation awarded $2.76 million in 2020 for 11 laboratory studies that are investigating whether there are underlying biological differences in the way people with CF may be infected by or respond to the novel coronavirus. The goal is to identify how people with CF may react differently or the same to the virus as those without CF by using cells in the lab to understand expected outcomes and improve future clinical care for those who contract the disease.
Fungal
There is a tremendous need to understand when to treat and how to treat people with CF who culture fungi. The CF Foundation is funding more research to help us understand the risks and benefits of treatment when an individual has a positive fungal culture. The Foundation is designing what will be the largest ever clinical study for Aspergillus in CF. The study, which is being supported by pilot studies in 2022, is expected to launch in 2023.
Stenotrophomonas maltophilia and other gram-negative bacteria
The Foundation is seeking to boost research efforts on other gram-negative bacteria, such as Achromobacter xylosoxidans and Stenotrophomonas maltophilia, as these were an identified area of need by the CF community and the Infection Research Steering Committee. Since the launch of the initiative, three new academic projects focused on understanding and treating Stenotrophomonas maltophilia have been initiated.
Driving Continued Progress
The Foundation will continue to seek and fund innovative science including both industry drug development programs and academic research. Companies should refer to Industry Funding Opportunities for information on how to apply for funding. If you are a researcher looking for upcoming funding opportunities, please refer to the Funding Opportunities page, which is regularly updated.
We welcome community input to inform our future priorities. If you are a person with CF or if you have a family member with CF and would like to be involved in setting research priorities or reviewing proposals submitted by CF researchers, please sign up to participate in Community Voice. Numerous opportunities to provide your input on Foundation activities are offered throughout the year.