I have always loved the excitement of the world's largest gathering of cystic fibrosis clinicians and researchers, known as the North American Cystic Fibrosis Conference (NACFC). Nearly 5,000 clinicians and researchers have been taking part in this year's conference in Nashville, Tenn., from Oct. 31-Nov. 2 where they have been learning more about the latest advances in CF research, care, and drug development.
This year has been even more exhilarating with the approval of the triple-combination modulator, the 30th anniversary of the crucial discovery of the gene that causes CF, and the Oct. 30 announcement of the Foundation's Path to a Cure initiative. These milestones provide us with an important opportunity to look ahead at what's next in CF.
The opening to the first plenary featured a tribute to the Foundation's president and CEO Preston Campbell, who will be retiring at the end of the year. Many remarked on Preston's commitment to leading with both his head and his heart throughout his 32 years of working in CF, and mentioned his signature line, “what will this mean for people with CF?”
“Scientific breakthroughs are happening more frequently, giving us new exciting paths to finish our journey,” Campbell said in an emotional speech. “Don't stop dreaming of a day when our journey is over, and all people with CF can say, 'I used to have CF'.”
Michael BoyIe, MD, the Foundation's current senior vice president of therapeutics development -- who will become president and CEO of the Foundation on Jan. 1 -- presented a compelling vision of the future, declaring we are about to enter the most transformative time in the history of CF and that our most important and our most challenging work is ahead of us.
Mike introduced Bill Skach, MD, senior vice president of research affairs at the Foundation, who outlined the Path to a Cure, the Foundation's $500 million research initiative to deliver treatments for the underlying cause of the disease and a cure for every person with cystic fibrosis. Bill explained how the Foundation is challenging academia and industry to accelerate the pace of progress in CF drug discovery and development.
“If the opportunities are there, we will even exceed our $500 million goal. This is not a limit. This is our target,” Bill said. “I'm absolutely convinced that this is a seminal moment in CF. It is rare that we have such an opportunity, and we need to seize that opportunity. We have the vision. We have the resources. And we have the determination.”
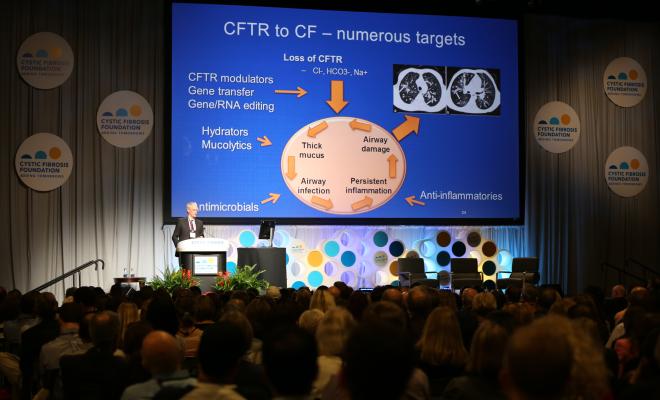
Marie Egan, MD, vice chair for research, Department of Pediatrics, Yale School of Medicine, then provided a deep dive into the science in her plenary talk, “Emerging Technologies That Will Lead to CFTR Restoration in All People With CF.” Dr. Egan discussed emerging, cutting-edge therapies, including novel approaches for people with nonsense mutations, as well as the challenges and opportunities that lie ahead as we work to develop novel genetic-based therapies.
In the second plenary, “Entering the Era of Highly Effective Modulator Therapy,” Jane Davies talked about the evolution of CFTR modulators and the potential benefits of introducing them to younger individuals. She also mentioned a new clinical trial called PROMISE that will study the short- and long-term effects of the new triple-combination therapy on the overall health of people with CF.
In addition, she highlighted a future study called SIMPLIFY that will examine reduced treatments in people with CF after they start highly effective modulators. Dr. Davies said treatment burden was the No. 1 research priority of people with CF in Europe, and I know it rated highly in surveys that have been conducted by the Foundation in the U.S.
Out of the many highlights from the first two plenaries, it was nice to end on a high note -- literally -- with a song from Dr. Francis Collins, the director of the National Institutes of Health and one of three researchers who co-discovered the CFTR gene in 1989. This breakthrough charted the path for the CF scientific community.
“We did dream back in 1989 that that discovery would have consequences. We didn't know what that pathway would look like. We didn't know how long the journey might be. It is remarkable to be able to be here in Nashville today and to say for 90 percent, we have arrived at a remarkable milestone,” Collins said, before singing a song that he wrote, “Dare to Dream.” “I am confident that we're not done yet. We're just getting started.”