This is an exciting time for cystic fibrosis research, with more than two dozen potential drugs being tested in clinical trials.
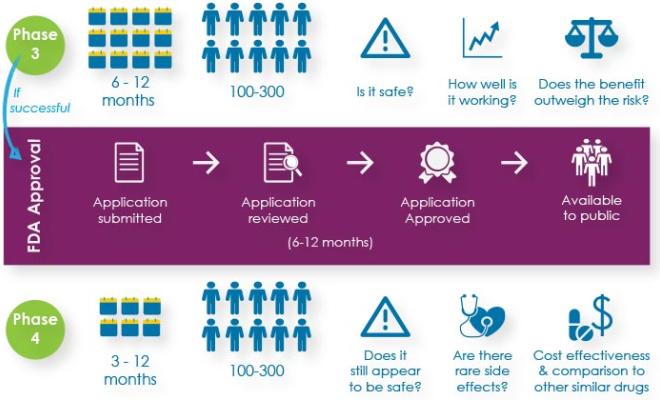
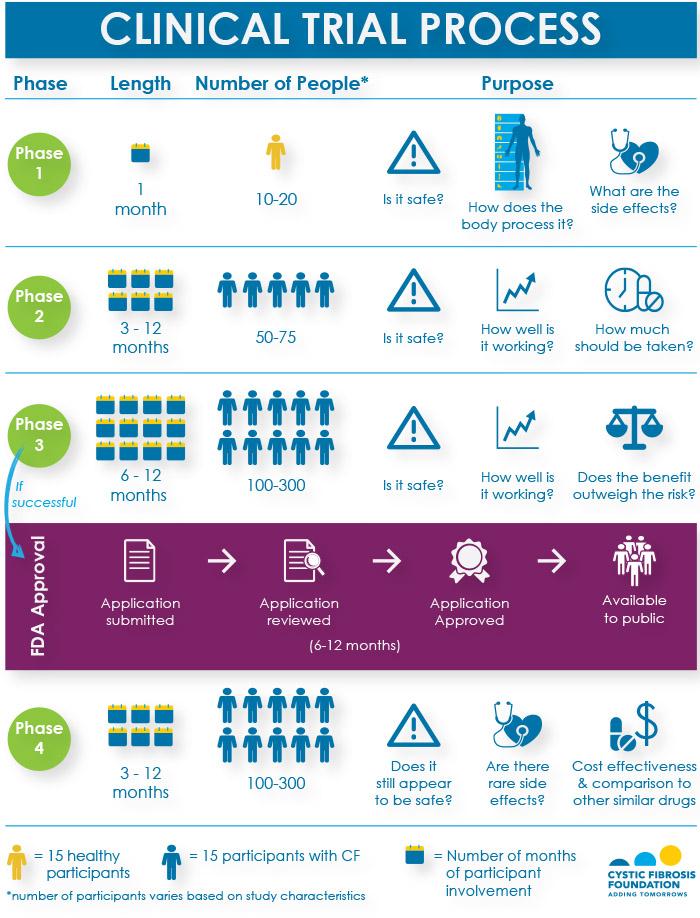
But in order for any new drug to become available to the public, it must pass through three phases of clinical trials to show that it is safe and effective in treating the disease, and receive approval by the U.S. Food and Drug Administration (FDA). If the FDA approves the drug, it will continue to be monitored for safety and effectiveness in what is known as a Phase 4 study.
Here's a breakdown of the questions that researchers try to answer, the number of participants needed and the time per participant for each phase of research.
Because there are so many promising new research opportunities, as many people with CF as possible are needed to participate in clinical trials.
Check out our Drug Development Pipeline to find out where new drugs are in the clinical trial process.